Laurence partnered with Cancer Research UK to raise funds for cervical cancer research.
In November 2023, England’s National Health Service announced a target to eliminate cervical cancer by 2040.
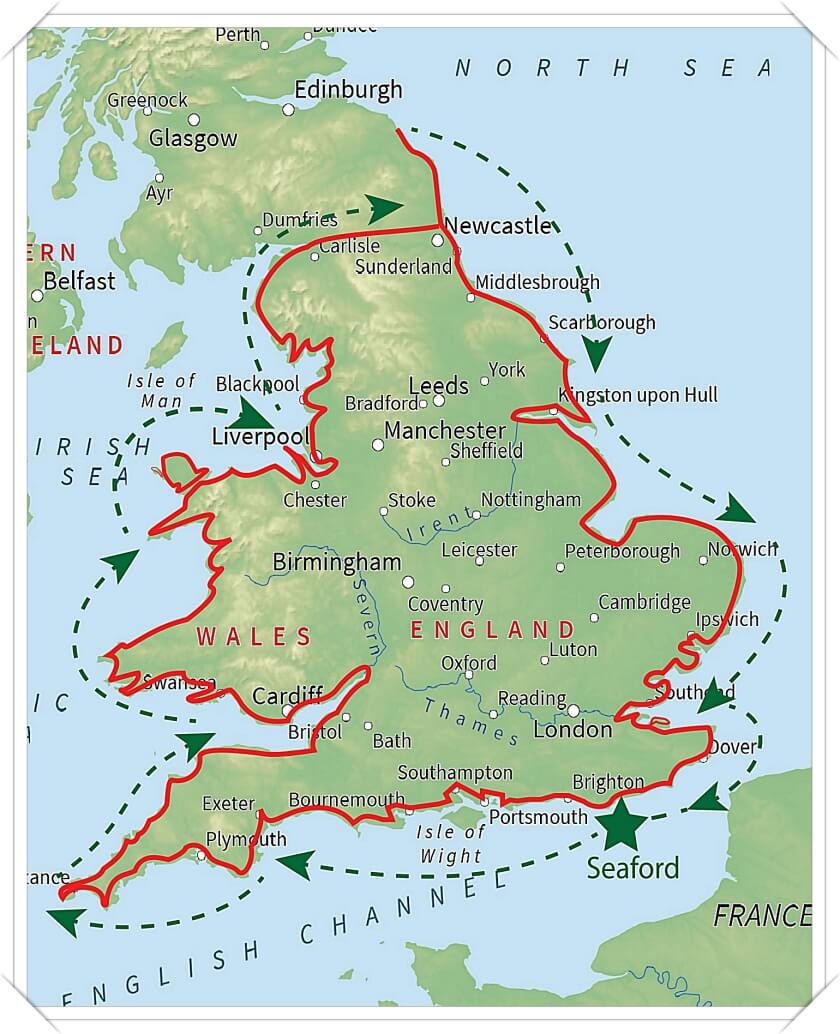
Laurence Carter: 3500 to end itI really enjoyed meeting Laurence Carter - such a positive man on a mission to stop others from suffering the pain he has endured. He's walking 3,500 miles around the coast of England and Wales in a year and trying to encourage more women to have their smear test on time, after his wife of 25 years died from cervical cancer. Good luck with the rest of your journey Laurence.
Posted by Fiona Marley Paterson ITV Border on Tuesday, February 12, 2019












































































